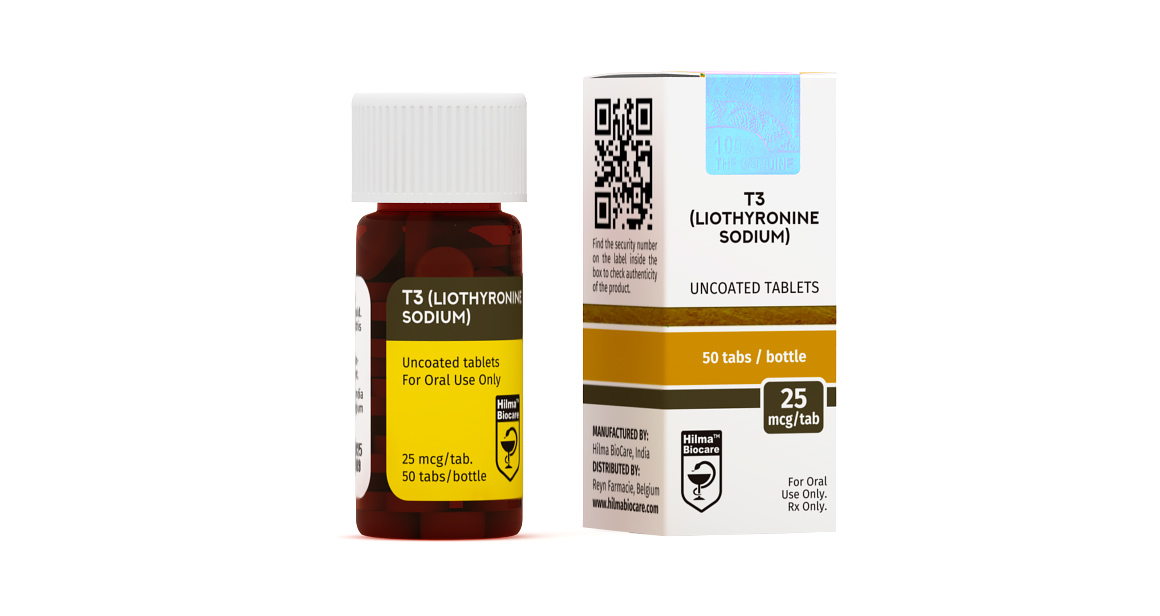
T3, LIOTHYRONINE SODIUM
Strength: 25 mcg
Molecular Formula: C15H11/3NNAO4
Molecular Weight: 672.96 g/mol
Active Ingredient: Liothyronine Sodium
CAS number: 55-06-1
Dosage Form: Tablet
Route:Oral
Market Status: Prescription
Company:Hilma Biocare
DESCRIPTION
Liothyronine sodium (T3) is not an anabolic steroid. It’s a synthetic (man-made) version of
one of the two hormones made by the thyroid gland, triiodothyronine. It is used for treating
individuals who are hypothyroid (do not produce enough thyroid hormones). Thyroid
hormones increase the metabolism (activity) of all cells in the body. In the fetus, newborn
infant and child, thyroid hormones promote growth and development of tissues. In adults,
thyroid hormones help to maintain the function of the brain, the use of food by the body, and
body temperature. Bodybuilders are particularly attracted to this drug for its ability to burn off
body excess fat. Most often utilized during contest preparation, one can greatly decrease the
amount of stored fat without being forced to severely restrict calories. To this end
liothyronine sodium is commonly used in conjunction with Clenbuterol and can produce
extremely dramatic results.
CLINICAL PHARMACOLOGY
Since liothyronine sodium (T3) is not firmly bound to serum protein, it is readily available to
body tissues. The onset of activity of liothyronine sodium is rapid, occurring within a few
hours. Maximum pharmacologic response occurs within 2 or 3 days, providing early clinical
response. The biological half-life is about 2-1/2 days. T3 is almost totally absorbed, 95
percent in 4 hours. The hormones contained in the natural preparations are absorbed in a
manner similar to the synthetic hormones. Liothyronine sodium (T3) has a rapid cutoff of
activity which permits quick dosage adjustment and facilitates control of the effects of
overdosage, should they occur.
INDICATIONS
Liothyronine is used to treat hypothyroidism (low production of thyroid hormone) in adults
and children. Prolonged hypothyroidism can result in a condition called myxedema in which
patients develop swollen lips, thickened nose, and unusual deposits of material in the skin
that are dry and waxy. These deposits also may appear in body tissues other than the skin.
Liothyronine also is used for suppressing production of thyroid stimulating hormone in
patients with goiters and for testing how well the thyroid gland is functioning.
ADVERSE REACTIONS
Adverse reactions, other than those indicative of hyperthyroidism because of therapeutic
overdosage, either initially or during the maintenance period are rare (see overdosage). In
rare instances, allergic skin reactions have been reported with Liothyronine Sodium Tablets,
USP.
CONTRAINDICATIONS
Thyroid hormone preparations are generally contraindicated in patients with diagnosed but
as yet uncorrected adrenal cortical insufficiency, untreated thyrotoxicosis and apparent
hypersensitivity to active or extraneous constituents. There is no well-documented evidence
from the literature, however, of true allergic or idiosyncratic reactions to thyroid hormone.
PRECAUTIONS
Thyroid hormone therapy in patients with concomitant diabetes mellitus or insipidus or
adrenal cortical insufficiency aggravates the intensity of their symptoms. Appropriate
adjustments of the various therapeutic measures directed at these concomitant endocrine
diseases are required.
DRUG INTERACTIONS
Oral Anticoagulants: thyroid hormones appear to increase catabolism of vitamin Kdependent clotting factors. If oral anticoagulants are also being given, compensatory
increases in clotting factor synthesis are impaired. Patients stabilized on oral anticoagulants
who are found to require thyroid replacement therapy should be watched very closely when
thyroid is started. If a patient is truly hypothyroid, it is likely that a reduction in anticoagulant
dosage will be required. Insulin or Oral Hypoglycemics: initiating thyroid replacement therapy
may cause increases in insulin or oral hypoglycemic requirements. Patients receiving insulin
or oral hypoglycemics should be closely watched during initiation of thyroid replacement
therapy. Digitalis: thyroid preparations may potentiate the toxic effects of digitalis. Thyroid
hormone replacement increases metabolic rate, which requires an increase in digitalis
dosage. Ketamine: when administered to patients on a thyroid preparation, this parenteral
anesthetic may cause hypertension and tachycardia. Use with caution and be prepared to
treat hypertension, if necessary. Vasopressors: thyroxine increases the adrenergic effect of
catecholamines such as epinephrine and norepinephrine. Therefore, injection of these
agents into patients receiving thyroid preparations increases the risk of precipitating
coronary insufficiency, especially in patients with coronary artery disease. Careful
observation is required.
DOSAGE AND ADMINISTRATION
Recommended starting dosage is 25 mcg daily. Daily dosage then may be increased by up
to 25 mcg every 1 or 2 weeks. Usual maintenance dose is 25 to 75 mcg daily. Usually, the
maximum dose for men shy of 100 micrograms per day, It’s not recommended to use more
than 50 mcg per day for women.
OVERDOSAGE
Signs and Symptoms: headache, irritability, nervousness, sweating, arrhythmia (including
tachycardia), increased bowel motility and menstrual irregularities. Angina pectoris or
congestive heart failure may be induced or aggravated. Shock may also develop. Massive
overdose may result in symptoms resembling thyroid storm. Chronic excessive dosage will
produce the signs and symptoms of hyperthyroidism.
Treatment Of Overdosage: dosage should be reduced or therapy temporarily discontinued if
signs and symptoms of overdosage appear. Treatment may be reinstituted at a lower
dosage. In normal individuals, normal hypothalamic-pituitary- thyroid axis function is restored
in 6 to 8 weeks after thyroid suppression.
PRESENTATION
Liothyronine Sodium (T3) 25 mcg uncoated tablets: 50 tablets in 1 bottle.
STORAGE
Store in a cool dry place between 15 – 25°C. Protect from light.
You can can find more in our online store hilmabiocare.co.uk














Reviews
There are no reviews yet.